Ion-controlled conformational dynamics in the outward-open transition from an occluded state of LeuT
By Chunfeng Zhao, Sebastian Stolzenberg, Luis Gracia, Harel Weinstein, Sergei Noskov, and Lei Shi.
Published in Biophysical Journal, 103 (5): 878-888 (2012). PMID: 23009837. PMCID: PMC3433624. Link to Pubmed page.
Project: The Transport Cycle in Neurotransmitter Uptake Systems
Abstract
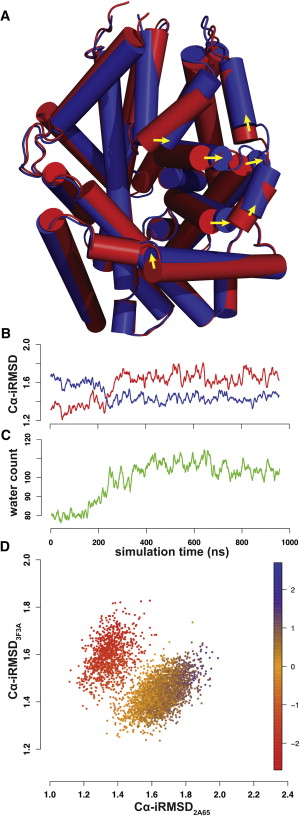
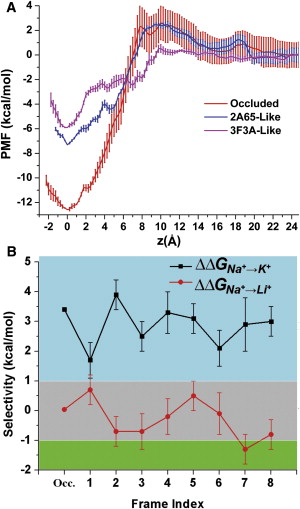
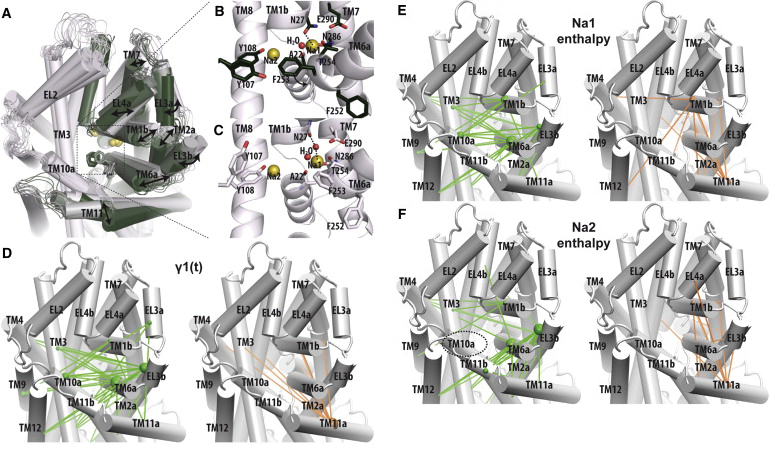
Neurotransmitter:sodium symporter (NSS) proteins are secondary Na+-driven active transporters that terminate neurotransmission by substrate uptake. Despite the availability of high-resolution crystal structures of a bacterial homolog of NSSs—Leucine Transporter (LeuT)—and extensive computational and experimental structure-function studies, unanswered questions remain regarding the transport mechanisms. We used microsecond atomistic molecular-dynamics (MD) simulations and free-energy computations to reveal ion-controlled conformational dynamics of LeuT in relation to binding affinity and selectivity of the more extracellularly positioned Na+ binding site (Na1 site). In the course of MD simulations starting from the occluded state with bound Na+, but in the absence of substrate, we find a spontaneous transition of the extracellular vestibule of LeuT into an outward-open conformation. The outward opening is enhanced by the absence of Na1 and modulated by the protonation state of the Na1-associated Glu-290. Consistently, the Na+ affinity for the Na1 site is inversely correlated with the extent of outward-open character and is lower than in the occluded state with bound substrate; however, the Na1 site retains its selectivity for Na+ over K+ in such conformational transitions. To the best of our knowledge, our findings shed new light on the Na+-driven transport cycle and on the symmetry in structural rearrangements for outward- and inward-open transitions.