Native chemical ligation at Asx-Cys, Glx-Cys: chemical synthesis and high-resolution X-ray structure of ShK toxin by racemic protein crystallography
By Bobo Dang, Tomoya Kubota, Kalyaneswar Mandal, Francisco Bezanilla, and Steve Kent.
Published in Journal of the American Chemical Society, 135(32):11911-9 on August 14, 2013. PMID: 23919482. PMCID: PMC3838204. Link to publication page.
Core Facility: Membrane Protein Expression/Purification
Abstract
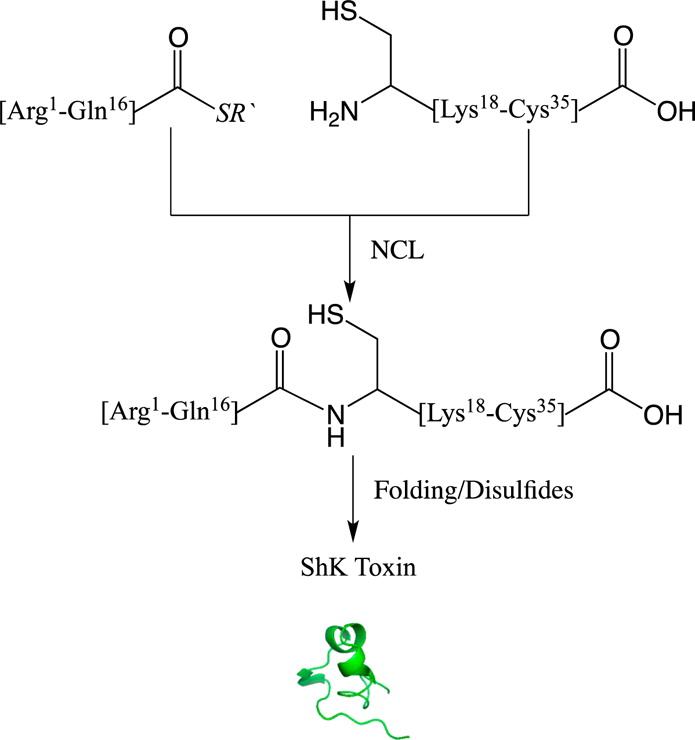
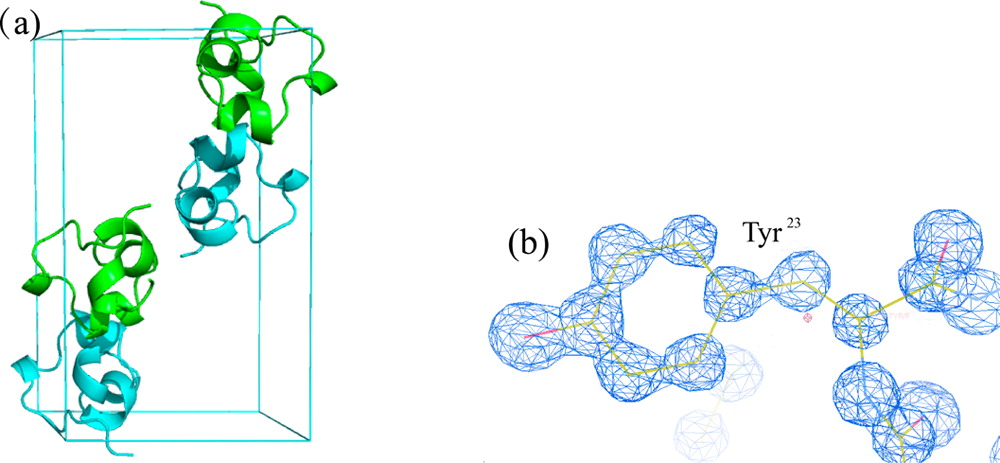
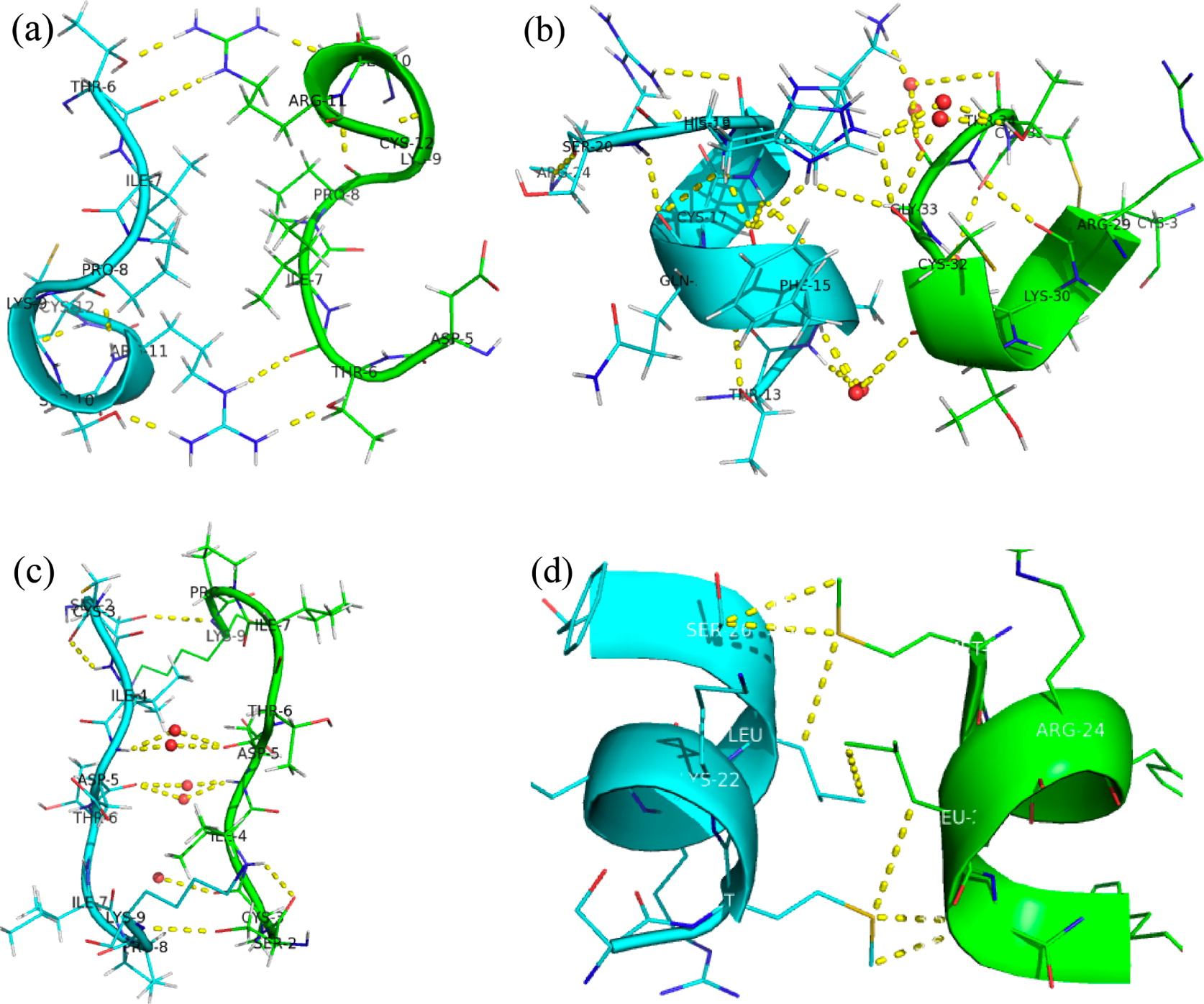
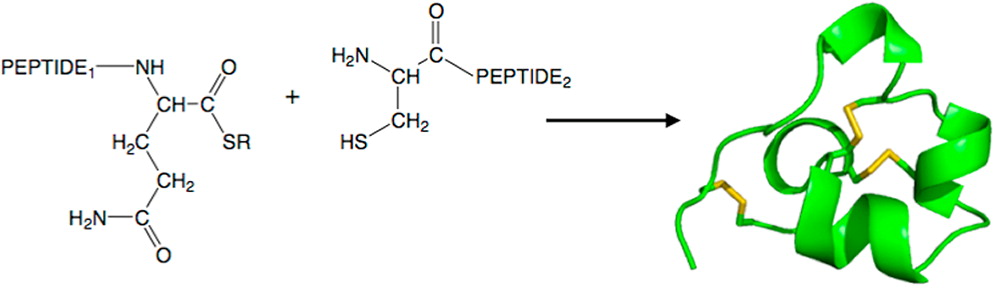
We have re-examined the utility of native chemical ligation at -Gln/Glu-Cys- [Glx-Cys] and -Asn/Asp-Cys- [Asx-Cys] sites. Using the improved thioaryl catalyst 4-mercaptophenylacetic acid (MPAA), native chemical ligation could be performed at -Gln-Cys- and Asn-Cys- sites without side reactions. After optimization, ligation at a -Glu-Cys- site could also be used as a ligation site, with minimal levels of byproduct formation. However, -Asp-Cys- is not appropriate for use as a site for native chemical ligation because of formation of significant amounts of β-linked byproduct. The feasibility of native chemical ligation at -Gln-Cys- enabled a convergent total chemical synthesis of the enantiomeric forms of the ShK toxin protein molecule. The d-ShK protein molecule was 50,000-fold less active in blocking the Kv1.3 channel than the l-ShK protein molecule. Racemic protein crystallography was used to obtain high-resolution X-ray diffraction data for ShK toxin. The structure was solved by direct methods and showed significant differences from the previously reported NMR structures in some regions of the ShK protein molecule.