Structural mechanism of voltage-dependent gating in an isolated voltage-sensing domain
By Qufei Li, Sherry Wanderling, Marcin Paduch, David Medovoy, Abhishek Singharoy, Ryan McGreevy, Carlos A Villalba-Galea, Raymond E Hulse, Benoît Roux, Klaus Schulten, Anthony Kossiakoff & Eduardo Perozo.
Published in Nature Structural & Molecular Biology 21(3) on March 28, 2014. PMID: 24487958. PMCID: PMC4116111. Link to publication page.
Project: Dynamics of Ion Permeation and Conformational Coupling in – KcsA. Core Facilities: Computational Modeling, sAB Generation and Crystallography.
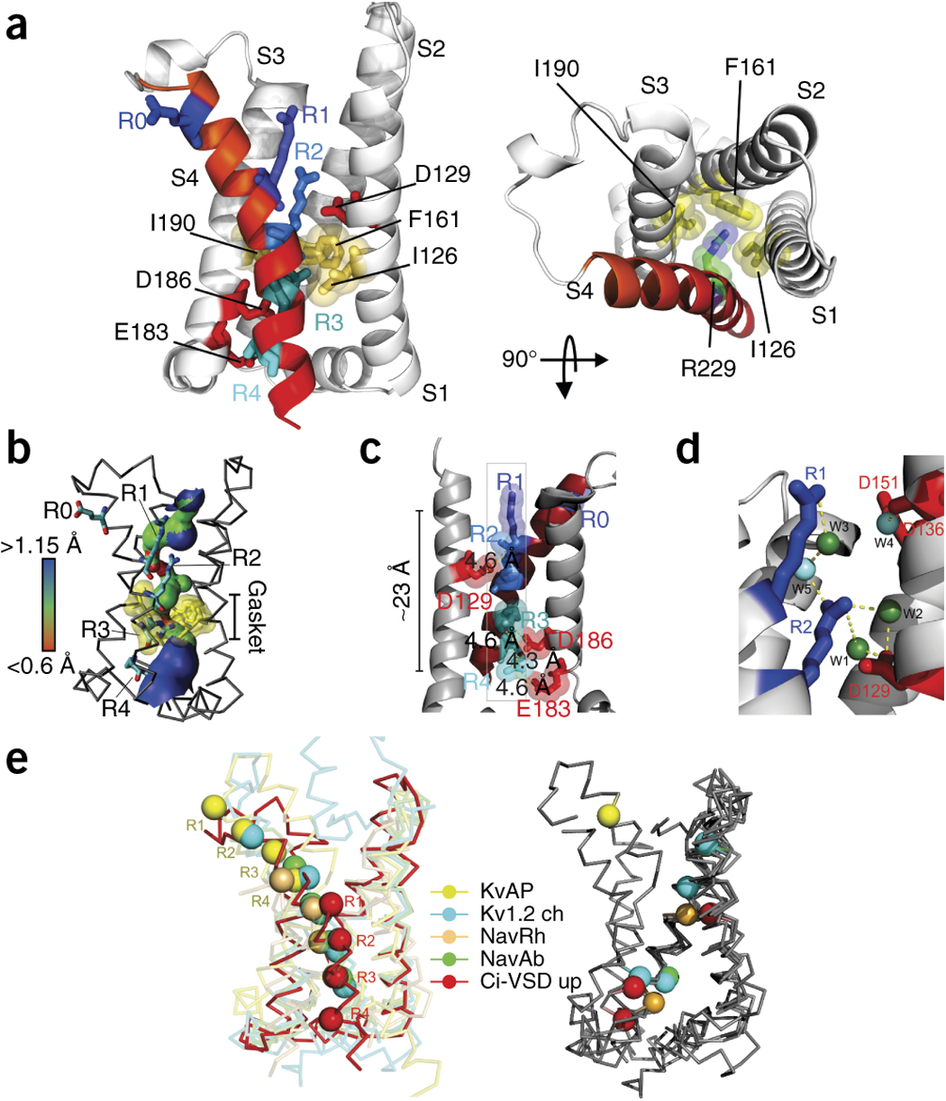
Figure 2. Nucleotide-binding pocket of PI4KIIα. (a) Side view (left; with extracellular side at top) and top view (right; from extracellular side) of the up conformation of Ci-VSD R217E in cartoon representation with S4 colored red. The four positive charges (R223, R226, R229 and R232) are depicted as sticks and colored in a series of blue shades. A well-defined hydrophobic gasket formed by I126, F161 and I190 with a gating charge R229 (R3) is shown. (b) Architecture of the internal and external vestibules, as calculated with HOLE56. The narrowest region is surrounded by the hydrophobic gasket at R3 (in yellow). (c) Structure showing all four gating charges forming a neat line perpendicular to the hydrophobic gasket. The negative countercharges (D129, D1; D186, D2 and E183, E3) in S1 and S3 are shown. (d) Structure showing stabilization of the top two arginines, R223 (R1) and R226 (R2) above the hydrophobic gasket, by hydrogen bonds with crystallographic waters (W) between the negative charges D129 and D151. (e) Left, heterogeneity of arginine positions within S4, highlighted with VSD alignment in reference to the Kv1.2 chimera (Kv1.2 ch, emerald). KvAP (yellow, r.m.s. deviation = 1.4 Å), NavRh (orange, r.m.s. deviation = 3.0 Å), NavAb (green, r.m.s. deviation = 1.1 Å) and Ci-VSD R217E (red, r.m.s. deviation = 1.1 Å). R.m.s. deviation is calculated with Cα of aligned region (S1, S2 and S3a) and indicates the general agreement of VSD scaffold. Right, heterogeneity of putative countercharge positions within the VSD scaffold. Only countercharges potentially involved in direct ion pairing with gating charges, on the basis of the crystal structures, are shown.
Abstract
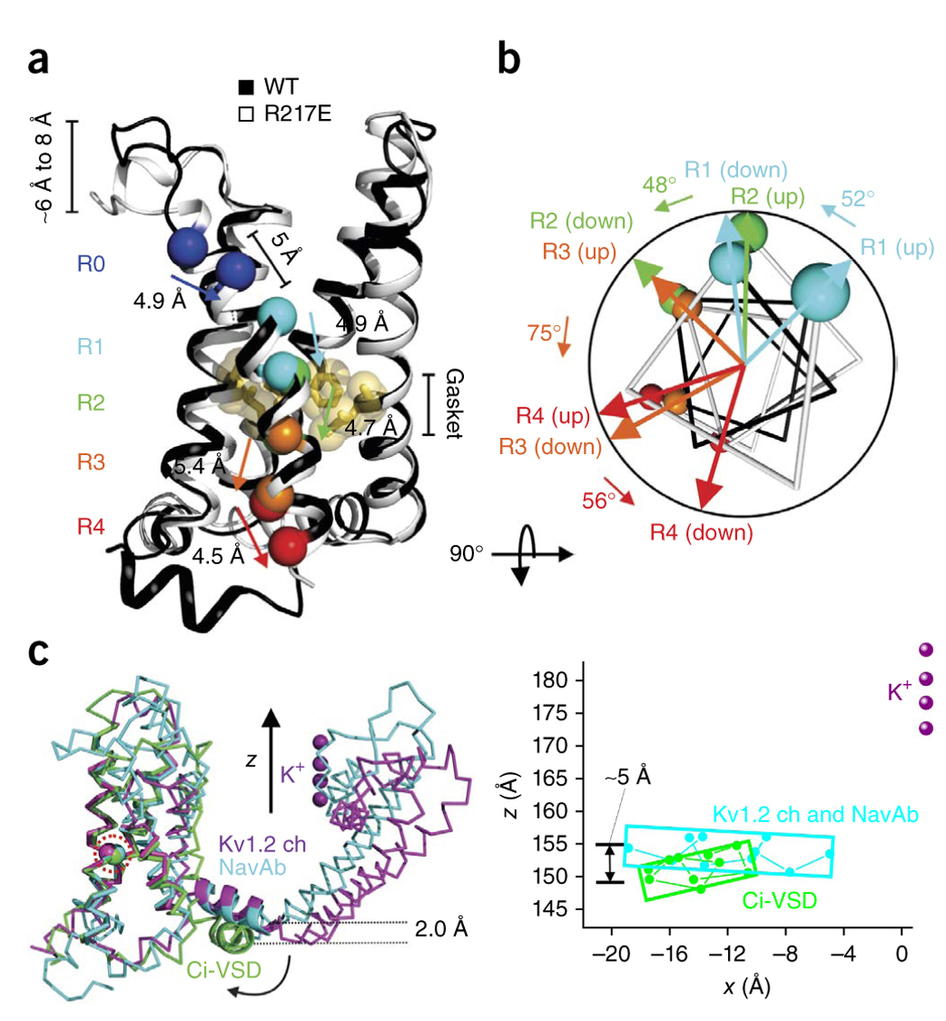
The transduction of transmembrane electric fields into protein motion has an essential role in the generation and propagation of cellular signals. Voltage-sensing domains (VSDs) carry out these functions through reorientations of positive charges in the S4 helix. Here, we determined crystal structures of the Ciona intestinalis VSD (Ci-VSD) in putatively active and resting conformations. S4 undergoes an ~5-Å displacement along its main axis, accompanied by an ~60° rotation. This movement is stabilized by an exchange in countercharge partners in helices S1 and S3 that generates an estimated net charge transfer of ~1 eo. Gating charges move relative to a ”hydrophobic gasket’ that electrically divides intra- and extracellular compartments. EPR spectroscopy confirms the limited nature of S4 movement in a membrane environment. These results provide an explicit mechanism for voltage sensing and set the basis for electromechanical coupling in voltage-dependent enzymes and ion channels.