Structure and permeation mechanism of a mammalian urea transporter
By Elena J. Levin, Yu Cao, Giray Enkavi, Matthias Quick, Yaping Pan, Emad Tajkhorshid, Ming Zhou.
Published in Proceedings of the National Academy of Sciences USA 109(28): 11194-9, on July 10, 2012. PMID: 22733730. PMCID: PMC3396522. Link to publication page.
Core Facility: Computational Modeling
Abstract
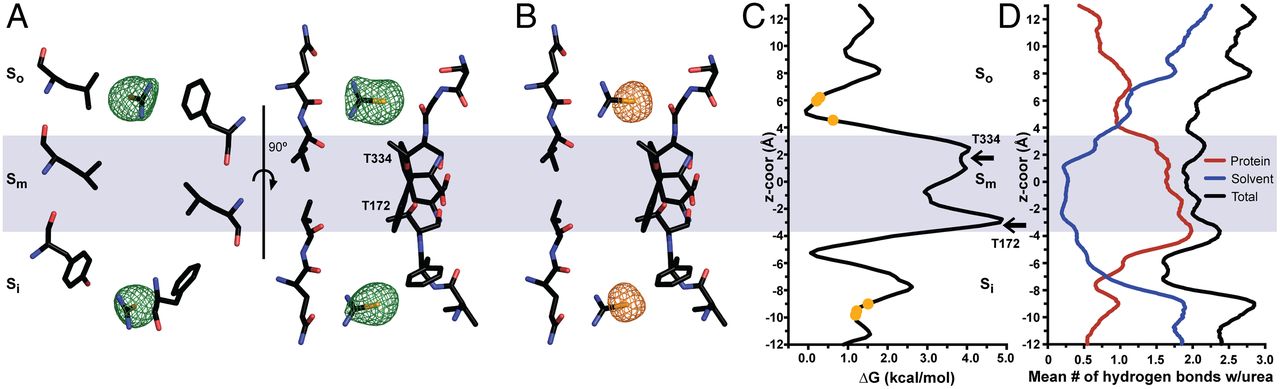
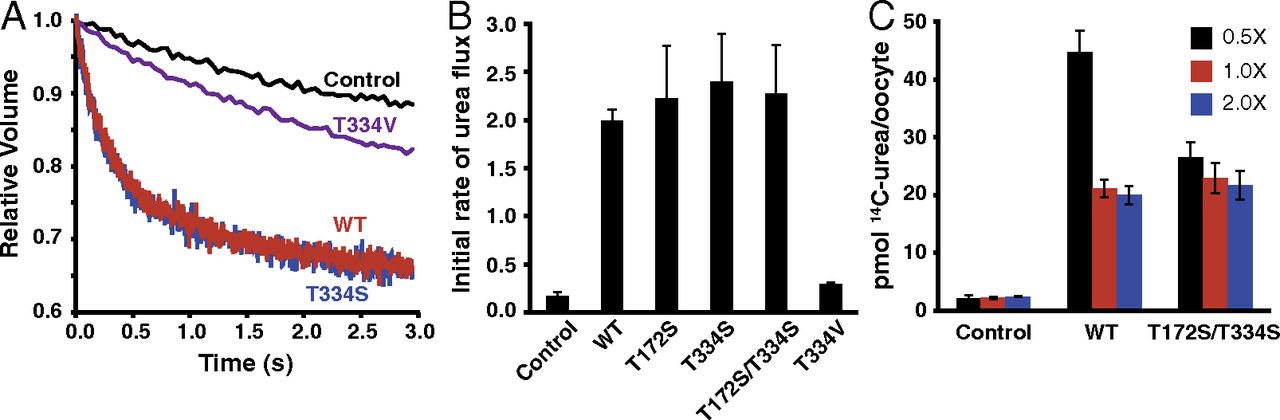
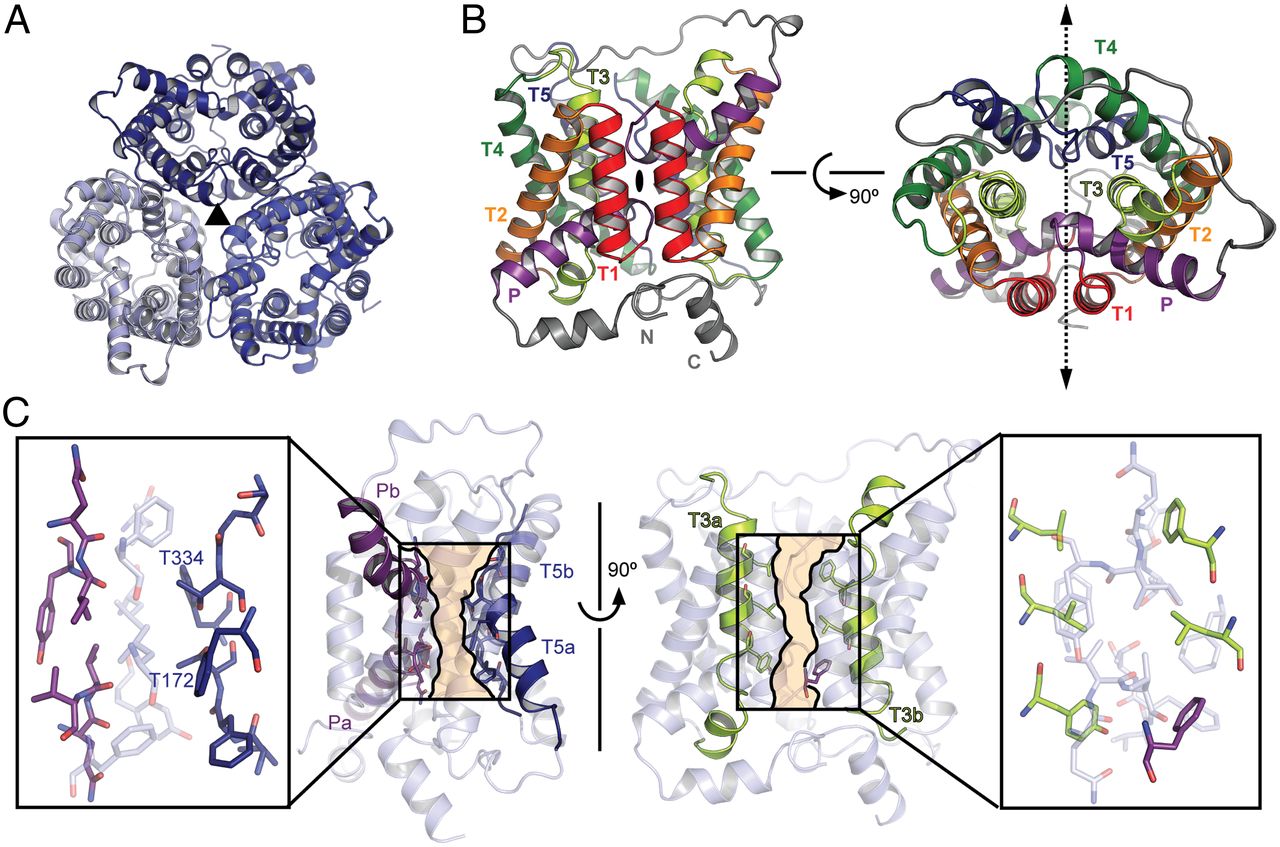
As an adaptation to infrequent access to water, terrestrial mammals produce urine that is hyperosmotic to plasma. To prevent osmotic diuresis by the large quantity of urea generated by protein catabolism, the kidney epithelia contain facilitative urea transporters (UTs) that allow rapid equilibration between the urinary space and the hyperosmotic interstitium. Here we report the first X-ray crystal structure of a mammalian UT, UT-B, at a resolution of 2.36 Å. UT-B is a homotrimer and each protomer contains a urea conduction pore with a narrow selectivity filter. Structural analyses and molecular dynamics simulations showed that the selectivity filter has two urea binding sites separated by an approximately 5.0 kcal/mol energy barrier. Functional studies showed that the rate of urea conduction in UT-B is increased by hypoosmotic stress, and that the site of osmoregulation coincides with the location of the energy barrier.